Snoring & sleep apnea can be the result of other illnesses or can increase the risk of developing other illnesses.

Diagnosis
Diagnosis
The difference between snoring and sleep apnea is how you feel during the day. Sleep apnea can be a serious disorder and can be associated with other serious illnesses such as heart disease, stroke, neurological disease, spinal or brain stem injury.
- Examinations:
- A physician will examine the throat, mouth, and nose for extra tissue or abnormalities. They will also check your blood pressure. If applicable, a physician may also require or review your data from a sleep center.
Symptoms
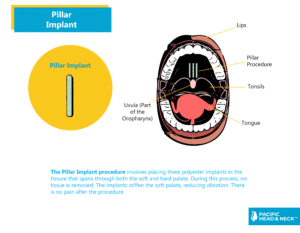
Treatment
Health Risks
Infographic